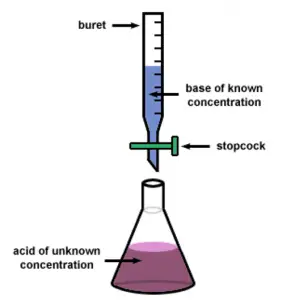
I am doing chemical titration for the very first time. What are the different experimental error chemistry student faces during titration? Reply ASAP.
Different Experimental Error Chemistry Student Faces During Titration

There are various types of errors that can make titration result deviate from the actual reading.
- First, there is the intrinsic error of the process – endpoint is not identical to the equivalence point, and color changes of the indicators are not instant. This may be due to the wrong choice of indicator, or negligence during titration.
- Then, there can be errors that are caused due to volumetric glass in These can be adjusted by the careful calibration of the glass. If du some reason calibration can’t be done, we can minimize the errors using A-class volumetric glassware.
- Misreading the color of the indicator near endpoint is probably the most common error. Not only color change is sometimes very slow, but different people have different sensitivity to colors. Distinguishing the perfect end point by the naked eye is actually quite difficult.
- Contamination of different chemicals can also change the end point.
- Improper filling of the burette can also give errors in volume reading. Thus the presence of air bubbles within the burette must be avoided.